List outlining eligibility for ‘Phase 2’ vaccination roll-out to be released soon
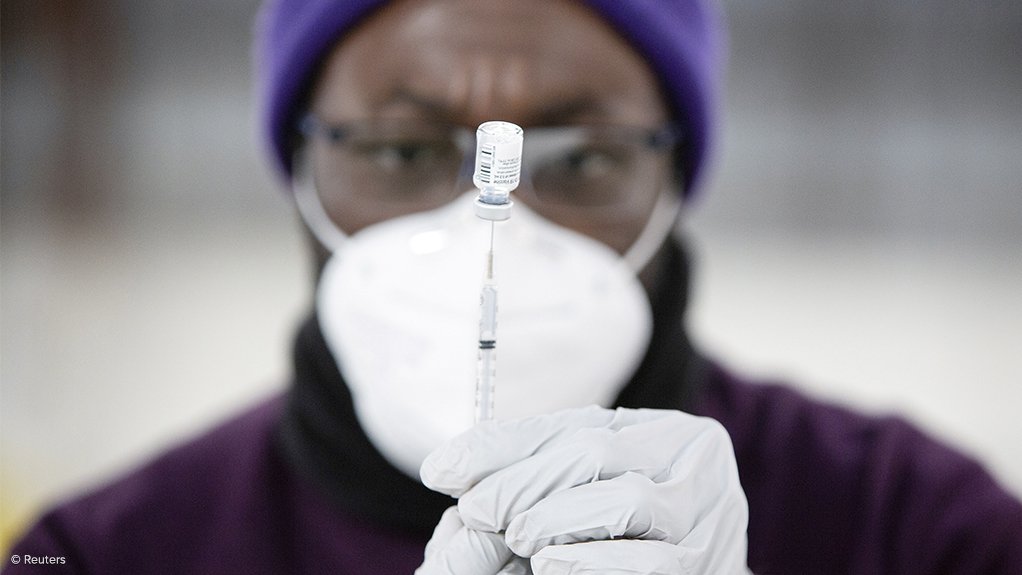
A list confirming which South Africans will be eligible for Covid-19 vaccination when the ‘Phase 2’ roll-out begins in late April or early May will be published in the “next week or two”, Department of Health deputy director-general Dr Anban Pillay reported on Friday.
Phase 2 will focus on the 16.6-million people falling into one of four categories: essential workers, persons in congregate settings, people over 60 years of age and individuals over 18 years with comorbidities.
Speaking during a webinar, Pillay reported that the Ministerial Advisory Committee on Covid-19 was currently finalising a list that would explicitly define each category.
He acknowledged that the final definitions, especially the classification of essential workers, was unlikely to satisfy all stakeholders, but stressed that priority would be given to those deemed to be at greatest risk of contracting severe disease or dying from Covid-19.
Business for South Africa’s (B4SA’s) Stavros Nicolaou said it was important for Phase-2 eligibility to be aligned with the known risk stratifications that had emerged since the onset of the pandemic so that the vaccination roll-out could target the most vulnerable groups ahead of the anticipated third wave and the onset of South Africa’s winter.
Research indicated that the elderly, for instance, were three times more likely to be hospitalised as a result of contracting Covid-19 and 15 times more likely to die.
The risks for people living with HIV and diabetes were also considerable, with a 50% higher hospital-admission risk and a 90% higher mortality rate.
Phase 2, once it began, would represent a major change in the pace and scale of South Africa’s vaccination effort, which was currently geared exclusively towards the vaccination of the country’s 1.25-million healthcare workers.
The programme was also currently only at the beginning stages of ‘Phase 1’ and was being facilitated through a Phase 3B open-label study of the single-dose Johnson & Johnson (J&J) vaccine to be administered to 500 000 frontline healthcare workers.
The study was initiated in mid-February after South Africa decided not to administer the AstraZeneca vaccine, following research showing that it was ineffective in preventing mild disease in patients who contracted the Covid-19 variant first found in South Africa and which had since become the dominant variant of the virus in the country.
South African Medical Research Council CEO Professor Glenda Gray said that the current slow pace of vaccination could be attributed largely to the fact that South Africa was still in a pre-roll-out phase, ahead of final registration of the J&J vaccine by the South African Health Products Regulatory Authority (SAHPRA).
Registration was expected to be imminent, however, while it was anticipated that Pfizer would be making an application to SAHPRA under Section 21 of the Medicines and Related Substance Act, through which unregistered medicines can be accessed.
Nicolaou estimated that government had already secured supply agreements with J&J and Pfizer to ensure that six-million people could be vaccinated by the end of June, taking into account the fact that the Pfizer vaccine involved a two-dose regimen.
B4SA had calculated that the country should aim to vaccinate eight-million people during the second quarter to dent the mortality curve, implying a shortage of doses for the period.
In the longer run, it had be confirmed that South Africa had secured enough vaccine doses to reach its goal of vaccinating 40-million adults above the age of 18 by early 2022.
Regardless of whether government could secure, during the second quarter, further doses there would still be a need to materially expand the network of vaccination sites and vaccinators to meet a daily vaccination tempo of between 120 000 and 250 000 people by the end of the second quarter.
That tempo would also need to be sustained for a prolonged period if South Africa was to meet its goal of vaccinating 67% of its population; currently considered the threshold for meeting community immunity.
Most South Africans, about 22.5-million in total, were expected to receive their vaccination during ‘Phase 3’, which was not expected to begin until the second half of the year.
Pillay said the vaccination sites would be expanded beyond hospitals and clinics to pharmacies, workplace-based facilities, general medical practitioners, as well as standalone facilities such as community halls and stadiums. Mobile vaccination solutions were also likely to be deployed.
All facilities would need to show that they could meet the minimum standards for operating as a vaccination site.
A consortium of chief information officers from large private companies was also working with government to improve the functioning of the Electronic Vaccine Data System (EVDS) through which individuals must register to secure a vaccination appointment.
The system was currently open for registration only by healthcare workers, but was being ‘stress tested’ to ensure that it could handle the large number of registrations that were likely to arise once Phase 2 registration was opened.
Nicolaou said that, while any registered entity could procure vaccines, B4SA supported centralised procurement by government during the initial stages of the vaccination drive and indicated that the private sector intended purchasing its initial doses from government.
He also stressed that South Africa had a cold-chain logistics infrastructure in place that could handle the vaccines, including the Pfizer vaccine, which required ultra-cold storage.
Article Enquiry
Email Article
Save Article
Feedback
To advertise email advertising@creamermedia.co.za or click here
Announcements
What's On
Subscribe to improve your user experience...
Option 1 (equivalent of R125 a month):
Receive a weekly copy of Creamer Media's Engineering News & Mining Weekly magazine
(print copy for those in South Africa and e-magazine for those outside of South Africa)
Receive daily email newsletters
Access to full search results
Access archive of magazine back copies
Access to Projects in Progress
Access to ONE Research Report of your choice in PDF format
Option 2 (equivalent of R375 a month):
All benefits from Option 1
PLUS
Access to Creamer Media's Research Channel Africa for ALL Research Reports, in PDF format, on various industrial and mining sectors
including Electricity; Water; Energy Transition; Hydrogen; Roads, Rail and Ports; Coal; Gold; Platinum; Battery Metals; etc.
Already a subscriber?
Forgotten your password?
Receive weekly copy of Creamer Media's Engineering News & Mining Weekly magazine (print copy for those in South Africa and e-magazine for those outside of South Africa)
➕
Recieve daily email newsletters
➕
Access to full search results
➕
Access archive of magazine back copies
➕
Access to Projects in Progress
➕
Access to ONE Research Report of your choice in PDF format
RESEARCH CHANNEL AFRICA
R4500 (equivalent of R375 a month)
SUBSCRIBEAll benefits from Option 1
➕
Access to Creamer Media's Research Channel Africa for ALL Research Reports on various industrial and mining sectors, in PDF format, including on:
Electricity
➕
Water
➕
Energy Transition
➕
Hydrogen
➕
Roads, Rail and Ports
➕
Coal
➕
Gold
➕
Platinum
➕
Battery Metals
➕
etc.
Receive all benefits from Option 1 or Option 2 delivered to numerous people at your company
➕
Multiple User names and Passwords for simultaneous log-ins
➕
Intranet integration access to all in your organisation